If you’re aiming for an A 1 in O-Level Chemistry in Singapore, the best tutor for you is someone who can explain MOE syllabus concepts clearly, drill you with exam-style questions, and is available when you’re actually stuck — not just once a week.
That usually means a mix of: a good human tutor or centre that fits your budget and schedule, plus an on-demand helper like Tutorly.sg’s 24/7 AI tutor to cover all the “I don’t get this” moments in between.
“Stuck on a question? See simple explanations that help you understand fast.”
👉 Give it a try and turn confusion into clarity in minutes.
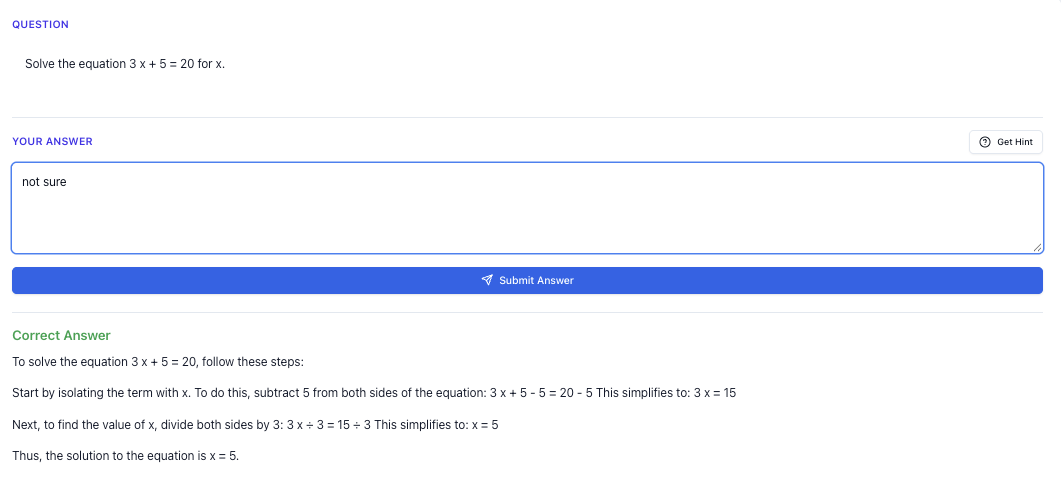
In this guide, I’ll walk you through how to choose the right O-Level Chemistry tutor in Singapore, how to study smarter for Paper 1 and Paper 2, and how to use tools like Tutorly to practise, check answers, and handle the harder variants that come out in the national exams.
Why O-Level Chemistry Feels So Hard (And What A Good Tutor Actually Fixes)
You already know this: O-Level Chemistry in Singapore isn’t just memorising facts.
You’re expected to:
- Understand concepts (like ionic vs covalent bonding),
- Apply them to new situations (novel experiment setups),
- And write answers in very specific exam language.
Common pain points I see from Sec 3–4 students:
- “I kind of understand in class, but when I see the question, I blank.”
- “I can do MCQ, but my structured answers always lose marks.”
- “Organic chem and mole concepts keep pulling my grades down.”
- “My school teacher goes fast and I’m too paiseh to ask again.”
A strong O-Level Chemistry tutor helps you:
- Fill concept gaps quickly .
- Translate understanding into exam answers (keywords, working, layout).
- Build question familiarity using past-year and “harder than school” questions.
- Plan a realistic study schedule around your other subjects.
This is also where an AI tutor like Tutorly.sg fits in nicely — it doesn’t replace your teacher, but it fills all the tiny gaps between lessons so you don’t fall behind.
Tutorly.sg has already been used by thousands of students in Singapore and has even been mentioned on Channel NewsAsia (CNA), so you’re not just trying some random website.
If you want to see how it works while reading this, you can try Tutorly instantly here.
Choosing The Right O-Level Chemistry Tutor In Singapore
You basically have three main options:
“Access more than 1000+ past year papers to practice”
👉 Start a paper today and test yourself like it’s the real exam.

- Private 1-to-1 tutor (home/online)
- Tuition centre (group classes)
- On-demand AI tutor (like Tutorly.sg)
Most students end up with a combination, not just one.
Rough price ranges in Singapore
These are typical ranges (not guarantees):
-
Private O-Level Chemistry tutor
- Undergrad / part-time: ~$1–$3 per hour
- NIE-trained / experienced full-time: ~$1–$3 per hour
- Famous “star” tutor: ~$1–$3+ per hour
-
Chemistry tuition centre (group)
- Small group : ~$1–$3 per month
- Big brand centres: can go up to ~$1–$3 per month
-
Tutorly.sg (AI tutor website)
- Free trial available; then usually a low monthly subscription that’s often less than a single 1.5-hour private tuition session.
Comparison: Private Tutor vs Tuition Centre vs Tutorly.sg
Here’s a quick comparison so you can see what fits your situation:
| Option | Price (rough) | Flexibility | Availability (time slots / urgency) |
|---|---|---|---|
| Private tutor | $1–$3/hour | Medium – fixed weekly slot, can reschedule with notice | Limited – depends on tutor’s schedule; hard to get last-minute help |
| Tuition centre | ~$1–$3/month | Low – fixed class times, makeup classes not always guaranteed | Low – only during class; maybe WhatsApp help if they offer |
| Tutorly (website) | Low monthly subscription (often <1 lesson) | Very high – you can log in anytime, short or long sessions | 24/7 – instant responses, even night before exam |
A smart approach for many Sec 3–4 students:
- Use a centre or private tutor for structured weekly teaching.
- Use Tutorly.sg for:
- Homework questions you can’t solve,
- Checking your answers to TYS,
- And revising weaker topics at your own pace.
You can open a new tab and get help now on Tutorly while you go through this guide.
Step-by-step Tutorial: Building Your O-Level Chemistry Study System
Instead of randomly doing assessment books, use this 5-step system. You can follow this with any tutor you choose — and strengthen it with Tutorly.
Step 1: Map out the MOE O-Level Chemistry syllabus
For pure Chemistry , key topic clusters include:
- Kinetic particle theory & states of matter
- Atomic structure & bonding
- Mole concept & stoichiometry
- Acids, bases & salts
- Redox, metals, electrolysis
- Energy changes
- Organic chemistry
- Experimental chemistry (planning, variables, errors)
Action:
- Print or save the official MOE syllabus (your school usually gives this).
- Highlight your weak areas honestly. Be specific: not “moles”, but “molar volume gas questions”.
Bring this to any tutor you’re considering. If they can’t quickly tell you how they’ll tackle your weak areas in a structured way, that’s a red flag.
With Tutorly, you can simply pick your level + Chemistry and ask targeted questions like:
“Explain redox in terms of electron transfer for O-Level, with one example question.”
Step 2: Build concept understanding (not just memorising answers)
A good O-Level Chemistry tutor should:
- Use simple analogies (e.g. “ions are like people who lost or gained items”),
- Draw clear links between topics (e.g. mole concept used in gas, solutions, titration),
- And constantly check your understanding with short questions.
Your job:
-
After each lesson (school or tuition), write 3–5 key ideas in your own words.
-
For example, after a lesson on ionic bonding:
- Ionic bonding is between metal and non-metal.
- Involves transfer of electrons to form ions.
- Ionic compounds have high melting points because of strong electrostatic forces.
- They conduct electricity only when molten or in aqueous solution.
Then, test yourself by asking:
“Can I explain this without looking at notes?”
If you get stuck, open Tutorly.sg and ask the AI tutor to re-explain the same concept in a simpler way or with more examples.
Step 3: Learn how to show working and use exam keywords
Understanding is not enough. O-Level examiners look for specific phrases and clear working, especially in Paper 2.
Examples:
-
Redox (in terms of electrons)
- Gain of electrons = reduction
- Loss of electrons = oxidation
Answering:
“In terms of electrons, explain why substance X is oxidised.”
You should write:
“Substance X is oxidised because it loses electrons.”
-
Electrolysis
Instead of writing:“Copper is formed at the cathode.”
Stronger answer:
“Copper ions, Cu²⁺, gain 2 electrons to form copper atoms at the cathode.”
A good tutor will constantly correct your phrasing. Tutorly can also help by:
- Checking your final answer,
- Then showing a full step-by-step solution so you can see the expected structure.
Step 4: Practise with increasing difficulty
You should move from:
- Basic school worksheet questions
- To assessment book / topical TYS questions
- Then to full TYS papers under timed conditions
Ask your tutor to:
- Start each new topic with easier questions to build confidence.
- Then quickly move you to harder variants .
On Tutorly, you can paste in any question (text) and:
- Check if your final answer is correct.
- If wrong, see a step-by-step explanation and compare to your own.
Step 5: Review mistakes properly (not just “oh, I see”)
After each test or practice paper:
-
Circle every question you lost marks on.
-
For each, write:
- What topic it belongs to.
- What type of mistake it was:
- Concept misunderstanding,
- Careless calculation,
- Misreading the question,
- Missing keywords.
-
Redo the question without looking at the solution.
If you still can’t get it, this is where:
- Your tutor should go through the thought process with you.
- Or you can paste the question into Tutorly and ask:
“Explain this step-by-step for O-Level standard.”
You can start building this habit now by trying a few questions with Tutorly here.
Exam Strategy Guide: Aiming For A 1 In O-Level Chemistry
Let’s talk about how to handle the actual O-Level exam, not just “study hard”.
Know your papers
For pure Chemistry (as of recent years – always confirm with your school):
-
Paper 1: MCQ (usually 40 marks)
- Tests wide content coverage.
- Many “trick” options based on common misconceptions.
-
Paper 2: Structured & Free Response (usually 80 marks)
- Longer questions, multi-part.
- Tests application, explanation, calculations, and experimental design.
Your strategy must cover both.
Paper 1 (MCQ) Strategy
-
Do not spend too long on any single question.
- Roughly, you have about 1–1.5 minutes per question.
- If stuck, circle it, move on, and come back later.
-
Elimination is your friend.
- Cross out clearly wrong options:
- E.g. “Ionic compounds conduct electricity as solids” → you know that’s false.
- Even if you’re unsure, narrowing down improves your odds.
- Cross out clearly wrong options:
-
Spot the “trap” patterns:
- Reversing definitions (e.g. oxidation vs reduction).
- Mixing up similar terms (e.g. endothermic vs exothermic).
- State symbols (s), (l), (g), (aq).
-
Do at least 5–10 full MCQ papers before prelims.
- Mark your score.
- For every wrong question, write down why you chose the wrong option.
Paper 2 Strategy
-
Scan the whole paper quickly first (1–2 mins).
- Identify:
- Calculation-heavy questions (moles, gas laws).
- Longer experimental planning questions.
- Identify:
-
Do the questions you’re confident in first.
- This builds momentum.
- You avoid getting stuck early and panicking.
-
Show clear working for calculations.
Use a consistent structure, for example:Write:
- Formula
- Substitution with units
- Final answer with correct unit and significant figures
-
Underline keywords in the question.
- E.g. “Describe and explain”, “State and explain”, “In terms of particles…”
- If they say “in terms of particles”, you must talk about particles, not just temperature.
-
Check for unit conversions.
- cm³ to dm³ .
- Minutes to seconds .
- These are easy marks to lose.
Time management by month (Sec 3 to O-Levels)
This is a rough guide; adjust based on your school’s pace.
-
Sec 3 (Term 3–4)
- Focus: Understand core topics (particles, atomic structure, bonding, moles).
- Action: Weekly practice on new topics + light revision of old ones.
-
Sec 4 (Term 1–2)
- Focus: New topics .
- Action: Start doing topical TYS for both Sec 3 and new topics.
-
Sec 4 (Term 3 – after mid-years)
- Focus: Full-paper practice + closing gaps.
- Action: 1–2 full papers per week, review mistakes thoroughly.
-
Prelims to O-Levels
- Focus: Exam stamina, speed, and high-yield topics.
- Action: Timed practice, targeted revision of weak topics.
If you’re reading this close to prelims or O-Levels and feeling behind, don’t panic. You can still benefit a lot from:
- A short-term intensive tutor,
- Plus nightly practice using Tutorly.sg’s AI tutor for specific questions.
Real-Life Scenario: Last-Minute Panic Before Prelims
Imagine this:
It’s 10.45pm on a Tuesday.
Your prelim Chemistry paper is tomorrow.
You’re stuck on a titration calculation from your school’s revision worksheet.
Your private tutor lesson was on Sunday.
Your tuition centre group chat is quiet.
Your parents are asleep. You’re stressed, tired, and tempted to just give up on that chapter.
This is exactly the kind of moment where an on-demand tool helps.
Instead of giving up, you:
- Open Tutorly.sg on your laptop.
- Select your level and Chemistry.
- Type the question (or the part you’re stuck on).
- Check your final answer.
- If it’s wrong, you read through the step-by-step solution, then try another similar question.
You go to sleep feeling at least 1 chapter more solid than before.
Over weeks and months, these “small saves” add up.
Worksheet Practice: From Standard To Hard O-Level Chemistry Questions
Let’s run through some practice styles you should be doing with your tutor and on your own. I’ll include harder variants similar to what often appears in O-Levels or top school prelims.
You can copy these into your own notes or try them directly with Tutorly.
Topic 1: Mole Concept & Stoichiometry
Standard question
A student burns 4.0 g of sulfur in excess oxygen to form sulfur dioxide, SO₂.
- Write a balanced chemical equation for the reaction.
- Calculate the number of moles of sulfur used.
- Hence, calculate the number of moles of SO₂ formed.
(Relative atomic mass: S = 32)
Outline of solution:
- Moles of S mol
- From equation, 1 mol S → 1 mol SO₂,
so moles of SO₂ = 0.125 mol
Harder variant (multi-step)
Calcium carbonate reacts with hydrochloric acid according to the equation:
2.50 g of CaCO₃ is added to excess HCl.
- Calculate the number of moles of CaCO₃ used.
- Calculate the number of moles of CO₂ produced.
- Calculate the volume of CO₂ produced at room temperature and pressure (r.t.p.), in cm³.
You should be able to:
- Convert mass → moles,
- Use mole ratio,
- Convert moles of gas → volume.
Try fully solving this yourself, then use Tutorly to:
- Check your final answer,
- And compare your working to the step-by-step solution it shows.
Topic 2: Acids, Bases & Titration
Standard question
25.0 cm³ of 0.100 mol/dm³ hydrochloric acid is exactly neutralised by 20.0 cm³ of sodium hydroxide solution.
The equation is:
Calculate the concentration of the sodium hydroxide solution in mol/dm³.
Key steps:
- Moles of HCl
- Use mole ratio to find moles of NaOH.
- Use to find concentration of NaOH.
Harder variant (with dilution)
A student prepares sodium hydroxide solution by dissolving 4.0 g of NaOH pellets in water to make 500 cm³ of solution.
25.0 cm³ of this solution is then completely neutralised by 30.0 cm³ of sulfuric acid, H₂SO₄.
The equation is:
- Calculate the concentration of the NaOH solution in mol/dm³.
- Calculate the concentration of the H₂SO₄ solution in mol/dm³.
This combines:
- Mass → moles → concentration,
- Then a titration calculation with a 2:1 mole ratio.
Again, solve it, then check with Tutorly to see each step clearly laid out.
Topic 3: Qualitative Analysis & Experimental Chemistry
Standard question
A student adds aqueous sodium hydroxide to a colourless solution and observes a white precipitate that is soluble in excess NaOH.
- Suggest the cation present.
- Write the ionic equation for the formation of the precipitate.
You should know:
- Al³⁺ gives a white ppt soluble in excess NaOH.
Harder variant (multi-observation)
A solution X is tested with aqueous sodium hydroxide and aqueous ammonia. The following observations are made:
- With aqueous NaOH:
- A light blue precipitate forms, soluble in excess to give a deep blue solution.
- With aqueous ammonia:
- A light blue precipitate forms, soluble in excess to give a deep blue solution.
- Identify the cation in solution X.
- Explain, in terms of complex ion formation, why the precipitate dissolves in excess reagent.
This tests:
- Your qualitative analysis table knowledge,
- Your ability to explain using correct terms (e.g. formation of a soluble complex ion).
Try forming a full exam-style answer, then paste your attempt into Tutorly and ask:
“Compare my answer with the ideal O-Level answer and show me the proper explanation.”
Topic 4: Organic Chemistry
Standard question
Ethene, C₂H₄, reacts with steam in the presence of a catalyst to form ethanol, C₂H₅OH.
- Name this type of reaction.
- State one condition needed for this reaction.
Expected:
- Addition reaction
- High temperature and catalyst (e.g. phosphoric acid on silica).
Harder variant (multi-part reasoning)
A hydrocarbon X has the molecular formula C₄H₈.
- Suggest whether X is likely to be saturated or unsaturated. Explain your answer.
- X is decolourised by bromine water in the absence of UV light. Write a chemical equation for this reaction.
- Another hydrocarbon Y has the formula C₄H₁₀. Explain why Y does not react with bromine water in the absence of UV light.
Here, examiners want:
- Correct use of “saturated/unsaturated”,
- A balanced equation,
- Clear explanation of reactivity vs non-reactivity.
You can test your answers and see a full model solution using Tutorly.sg.
Common Mistakes O-Level Chemistry Students In Singapore Make
Even strong students lose marks on the same few things. Knowing these helps you choose a tutor who actually targets them — and helps you use Tutorly more effectively.
1. Memorising without understanding
You might:
- Memorise the colour of precipitates,
- Memorise half-equations,
- But cannot explain why something happens.
Examiners test understanding by:
- Giving unfamiliar experiments,
- Asking you to predict results or explain in terms of particles / electrons.
Fix:
- Ask “why?” in every lesson.
- When using Tutorly, don’t just ask for final answers; ask:
“Explain why this happens in simple terms for O-Level.”
2. Weak on mole concept and basic math
Moles appear in:
- Stoichiometry,
- Gas laws,
- Concentration calculations,
- Titrations.
If your basic mole concept is shaky, everything else becomes painful.
Fix:
- Spend 1–2 weeks just drilling mole problems.
- Ask your tutor to start from simple mass–mole–volume conversions.
- On Tutorly, practise multiple similar questions until you stop making the same mistakes.
3. Not using proper exam keywords
For example:
- Saying “particles move faster” without mentioning kinetic energy.
- Saying “more collisions” without “more frequent effective collisions”.
These small things cost 1–2 marks each, which adds up.
Fix:
- Build a “keywords list” for each topic.
- Ask your tutor to highlight phrases you must use.
- When you see Tutorly’s solutions, pay attention to repeated phrases — these are usually exam-style.
4. Ignoring the “explain your answer” parts
Many students:
- State the correct answer,
- But skip explanation or write one vague line.
Example:
“Rate increases because temperature increases.”
This is too vague. A better O-Level answer:
“At higher temperature, particles have more kinetic energy, so more particles have energy equal to or greater than the activation energy, leading to more frequent effective collisions per unit time, so the rate of reaction increases.”
Fix:
- When you see “Explain”, think: cause → effect → link to concept.
- Practise writing full explanations and compare with Tutorly’s model answers.
5. Only practising “school level” questions
School worksheets can sometimes be:
- Too guided,
- Too similar to what you’ve already seen.
O-Level and top school prelim questions often:
- Combine multiple topics,
- Use unfamiliar contexts (e.g. new industrial processes),
- Require interpretation of graphs or tables.
Fix:
- Use a mix of:
- School worksheets,
- Assessment books,
- TYS ,
- And harder prelim papers if your tutor has them.
- Use Tutorly to break down
“Practice PSLE Science questions and get clear, step-by-step answers instantly.”
👉 Try a question now and see how fast you can improve.
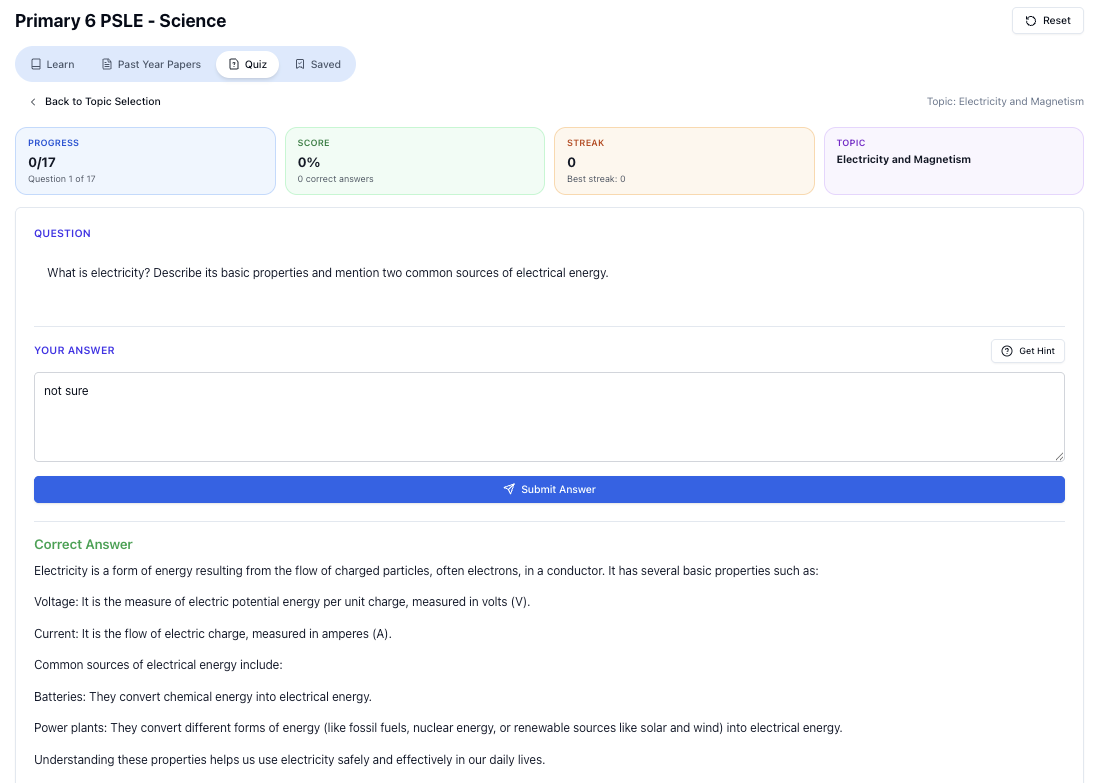
Ready to practise?
If you want a Singapore-focused AI tutor you can use immediately , try Tutorly here: