If you’re taking H 2 Chemistry in JC, you already know: it’s not “just another science subject”.
You’re juggling organic mechanisms, equilibrium, electrochemistry, energy cycles, and then trying to apply all of that in tricky, application-heavy A Level questions. On top of that, you still have GP, math, and maybe another H 2 science.
“Stuck on a question? See simple explanations that help you understand fast.”
👉 Give it a try and turn confusion into clarity in minutes.
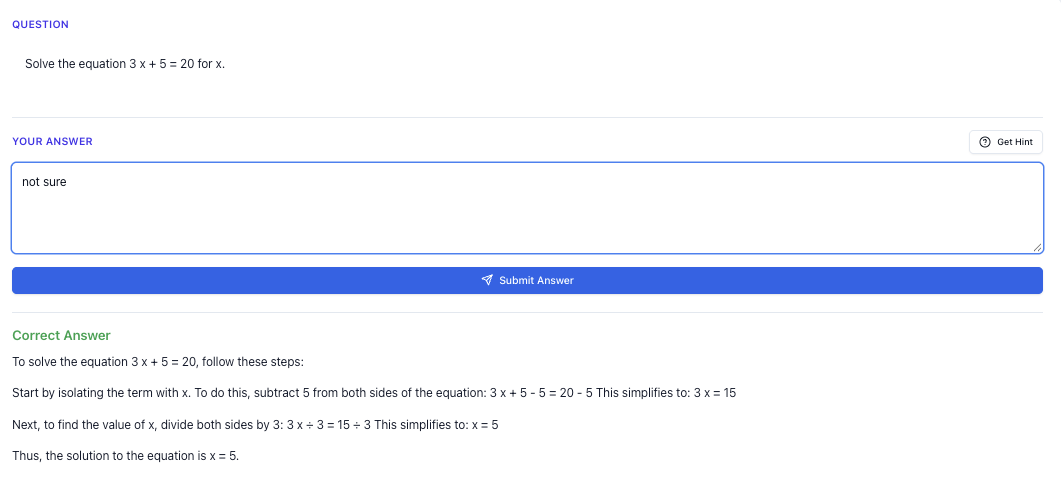
So when people talk about H 2 Chem tuition, what they really want is not “more content”. You already have lecture notes, tutorials, Ten Year Series (TYS). What you actually need is:
- Clear, step-by-step explanations when you’re stuck
- Targeted practice that feels like (or harder than) A Levels
- Fast feedback so you don’t keep repeating the same mistakes
- Exam strategies specific to the Singapore A Level H 2 Chemistry paper
That’s where focused H 2 Chem tuition and smart tools like Tutorly.sg come in.
Tutorly.sg is a 24/7 AI tutor website built specifically for Singapore students (Primary 1 to JC 2) and aligned to the MOE syllabus. It’s not a random overseas AI; it’s tuned for H 2 Chem topics, A Level command words, and local exam style. It’s also been mentioned on Channel NewsAsia (CNA) and used by thousands of students in Singapore, so you’re not exactly “experimenting” with something untested.
You can try the AI H 2 Chem tutor directly here:
👉 https://tutorly.sg/ai-tutor-singapore
In this guide, I’ll walk you through:
- How focused H 2 Chem tuition really boosts your A Level performance
- A step-by-step tutorial approach to mastering a topic
- A realistic exam strategy guide for Papers 1, 2, and 3
- How to do worksheet practice with hard variants, not just basic questions
- The common mistakes H 2 Chem students in Singapore keep making (and how to stop)
Why Focused H 2 Chem Tuition Matters (More Than “Just Studying Hard”)
You can mug the entire notes and still get stuck at C or B for H 2 Chem. The problem usually isn’t effort; it’s how you’re learning.
Focused H 2 Chem tuition (whether with a human tutor, school consults, or an AI tutor like Tutorly.sg) helps you:
-
Turn content into exam skills
You don’t just memorise “Le Chatelier’s Principle”. You learn:- How to predict direction of shift
- How to link it to or
- How to write a full, logical explanation worth 3–4 marks
-
Fill in topic gaps before they snowball
If your mole concept is weak, you will suffer in:- Energetics (enthalpy calculations)
- Equilibrium (Kc calculations)
- Acid–base (titration, pH calculations)
A good tutor or AI tutor can quickly diagnose: “Your issue is not equilibrium; it’s moles.”
-
Practise at the right difficulty
Many students stay stuck at:- Simple recall questions
- Basic structured questions
But A Level H 2 Chem loves: - Multi-step reasoning
- Cross-topic questions
Focused tuition pushes you into that zone before the exam.
-
Get instant, topic-specific help
You don’t want to wait 3 days for a consult slot just to ask, “Why is this nucleophile stronger?”
With Tutorly.sg, you can paste the question, and the AI tutor walks you through the reasoning step by step, any time, even at 1am.
If you’re already going for physical tuition, you can still use Tutorly.sg in between lessons to clarify doubts, drill MCQs, or check structured question answers.
Step-by-step Tutorial: Mastering a Typical H 2 Chem Topic
Let’s take a common pain point: Chemical Equilibrium .
“Access more than 1000+ past year papers to practice”
👉 Start a paper today and test yourself like it’s the real exam.

I’ll show you a step-by-step way to learn it, then show how you can use Tutorly.sg as your “on-demand tutor” at each step.
Step 1: Get the big picture first
Before diving into formulas, ask yourself:
-
What is equilibrium?
A dynamic state where forward and backward reactions occur at the same rate. -
What is ?
The equilibrium constant in terms of concentration; a measure of how far a reaction goes to completion. -
Why does it matter in A Levels?
Because they test:- Calculations (finding , concentrations)
- Conceptual shifts (how position of equilibrium changes)
- Links to Gibbs free energy, Le Chatelier, and sometimes acid–base systems
Use your lecture notes to get this overview. Don’t jump straight into TYS yet.
Step 2: Learn the core formulas and definitions properly
For equilibrium, you must be fluent with:
-
General expression, e.g. for :
-
What is not included (pure solids, pure liquids in heterogeneous equilibrium).
-
Relationship between and :
- : reaction shifts forward
- : reaction shifts backward
At this stage, you can use Tutorly.sg like this:
“Explain the difference between and for H 2 Chemistry, and give me 3 short MCQs to test my understanding.”
The AI tutor will generate questions aligned to H 2 style and explain each answer.
Step 3: Practise simple calculation questions
Start with:
- Finding from equilibrium concentrations
- Given and initial concentrations, find equilibrium concentrations using ICE tables
Example:
At 700 K, hydrogen and iodine react to form hydrogen iodide:
Initially, 1.00 mol of and 1.00 mol of are placed in a 1.00 dm container. At equilibrium, 0.20 mol of remains. Calculate .
Work through this yourself. Then, on Tutorly.sg, you can:
- Paste the question
- Type your final answer
- Let the AI tutor:
- Check your final answer
- Show you the full working step-by-step (even if your method was different)
You’ll see how to systematically set up the ICE table and avoid careless errors.
Step 4: Move to conceptual, explanation-type questions
Once you’re okay with calculations, move to “explain” questions:
- “Explain, in terms of Le Chatelier’s Principle, how an increase in pressure affects the equilibrium yield of ammonia in the Haber process.”
- “Explain why a catalyst does not change the value of .”
These are the questions where students lose marks because of vague phrasing.
Here’s how to use Tutorly.sg effectively:
-
Type your own full answer in proper exam style.
-
Ask:
“This is my answer to an H 2 Chem equilibrium question. Show me how the full-mark answer would be written, and highlight how mine is different.”
-
Compare:
- Are you missing key phrases like “position of equilibrium”, “partial pressures”, “rate of forward and backward reactions”?
- Are you mixing up “equilibrium constant” vs “equilibrium position”?
This is how you turn okay answers into A-grade, full-mark answers.
Step 5: Combine with other topics (cross-topic practice)
A Level questions love to mix:
- Equilibrium + acids and bases
- Equilibrium + solubility product
- Equilibrium + energetics
Example:
A saturated solution of a sparingly soluble salt AB has a concentration of mol dm.
(a) Calculate the solubility product, , of AB.
(b) Hence, explain and calculate what happens to the solubility if a solution of a common ion, e.g. ANO, is added.
Use Tutorly.sg to:
- Get variations of this question
- Ask for harder follow-up parts
- See step-by-step calculations and explanation
This is where focused H 2 Chem tuition really helps: you’re no longer just doing “equilibrium questions”; you’re doing A Level-style integrated questions.
Exam Strategy Guide for H 2 Chemistry (A Levels, Singapore)
Now let’s shift to exam strategy. Content is one thing; exam performance is another.
H 2 Chem A Levels usually has:
- Paper 1 (MCQ) – 30 questions, speed + accuracy
- Paper 2 (Structured) – shorter questions across many topics
- Paper 3 (Free-response) – longer, more integrated questions
Here’s how focused H 2 Chem tuition and tools like Tutorly.sg can help you tackle each.
1. Strategy for Paper 1 (MCQ)
Paper 1 is where many students lose easy marks due to:
- Misreading
- Rushing
- Weak conceptual understanding
Tactics:
-
Do topic-by-topic MCQs first
-
After finishing a topic (e.g. Redox), drill MCQs from that topic.
-
On Tutorly.sg, you can request:
“Give me 10 H 2 Chem MCQs on redox, similar to A Level standard, and explain each answer.”
-
-
Identify your weak topics by error patterns
- If you keep getting organic mechanisms wrong, that’s a signal.
- Use your tuition time or AI tutor time to re-learn that topic before doing more MCQs.
-
Simulate full Paper 1 conditions
- 30 questions, 1 hour
- No checking answers in between
- After finishing, then use Tutorly.sg to:
- Check each answer
- Ask for explanation for any you got wrong
2. Strategy for Paper 2 (Structured)
Paper 2 tests:
- Breadth of content
- Ability to write concise, accurate answers
- Calculations + short explanations
Tactics:
-
Practise writing in “mark scheme language”
- Instead of “the reaction goes more to the right”, write “the position of equilibrium shifts to the right”.
- Instead of “more products formed”, write “the yield of [product] increases”.
-
Time management
- Don’t spend 10 minutes stuck on one 4-mark question.
- If stuck, write something reasonable, leave a line, move on.
-
Use Tutorly.sg as a marking partner
-
After doing a full structured question:
-
Type your answer
-
Ask:
“This is my answer to a H 2 Chem structured question. Show me a full-mark answer.”
-
-
Compare and adjust your style.
-
3. Strategy for Paper 3 (Free-response)
Paper 3 is usually the most painful for students because:
- Questions are long
- They span multiple topics
- They require sustained concentration
Tactics:
-
Don’t fear long questions – break them
- Treat each part (a), (b), (c) as mini-questions.
- Underline what each part is actually asking (definition? calculation? explanation?).
-
Expect cross-topic integration
For example:- Organic + spectroscopy
- Energetics + bond energies + equilibrium
- Redox + electrochemistry + cell potential
-
Train with harder-than-exam questions
-
If you only do standard TYS, Paper 3 may still feel overwhelming.
-
With Tutorly.sg, you can request:
“Give me a hard H 2 Chem free-response question that mixes energetics and equilibrium, similar or slightly harder than A Levels.”
-
Then practise under timed conditions and review the model solution.
-
-
Use step-by-step solutions to learn patterns
- How does the solution start? (Define terms? Draw a diagram? Write the equation?)
- What structure do full-mark explanations follow?
Over time, you’ll start recognising patterns in how A Level free-response questions are set and how they’re marked.
Worksheet Practice: From Basic to Hard A Level Variants
You can’t “feel ready” for H 2 Chem without serious practice.
Here’s how to structure your worksheet practice, with examples of hard variants that are closer to A Level difficulty.
1. Build your own mini-worksheets by topic
Pick a topic, e.g. Organic Chemistry – Nucleophilic Substitution.
Your worksheet should include:
-
Basic recall
- Define nucleophile
- State the difference between SN 1 and SN 2 mechanisms
-
Standard exam-style questions
- Draw the mechanism for the reaction of bromoethane with hydroxide ions in ethanol.
- Explain how the rate of SN 1 vs SN 2 depends on the nature of the substrate.
-
Hard variants (A Level style)
Example:(a) 2-bromopropane reacts with aqueous sodium hydroxide and with ethanolic sodium hydroxide.
(i) State the type of reaction that occurs in each case.
(ii) Predict the major organic product for each reaction and explain, in terms of mechanism, why different products are formed.
(b) A student proposes that the reaction with aqueous sodium hydroxide proceeds via an SN 2 mechanism. Explain why this is unlikely, with reference to the structure of 2-bromopropane and the conditions used.
When you’re done:
- Type your answers into Tutorly.sg
- Ask for a full worked solution
- Compare mechanisms, curly arrows, and explanations
You can repeat this for every key topic: Redox, Equilibrium, Acid–Base, Electrochemistry, Organic, Energetics, Kinetics.
2. Use “progressive difficulty” questions
For each topic, try this pattern:
- Level 1 – Simple
- Single-step calculation or one concept
- Level 2 – Medium
- Two-step calculation or explanation + calculation
- Level 3 – Hard
- Multi-part, cross-topic, or unfamiliar context
Example: Energetics
Level 1 (Simple):
The enthalpy change of combustion of ethanol is kJ mol. Calculate the heat released when 0.50 mol of ethanol is completely burnt.
Level 2 (Medium):
Using the following data, calculate the enthalpy change of reaction for:
Given:
- kJ mol
- kJ mol
- kJ mol
Level 3 (Hard, A Level style):
A student burns a sample of ethanol in a spirit burner to heat 200 g of water in a calorimeter. The temperature of the water rises from 25.0 °C to 42.0 °C.
(a) Calculate the heat absorbed by the water.
(b) If 0.60 g of ethanol was burnt, calculate the experimental enthalpy change of combustion of ethanol in kJ mol.
(c) The standard enthalpy change of combustion of ethanol is more exothermic than the student’s value. Suggest and explain two reasons for this difference.
Ask Tutorly.sg to:
- Mark your final numerical answers
- Show the full working and explanation
- Provide 2–3 even harder variants, e.g. involving Hess’ Law cycles or bond energy approximations
3. Practice hard variants for Paper 3-style questions
Here are some harder, realistic variants you can try (and then check with Tutorly.sg):
Hard Variant 1: Equilibrium + Kinetics
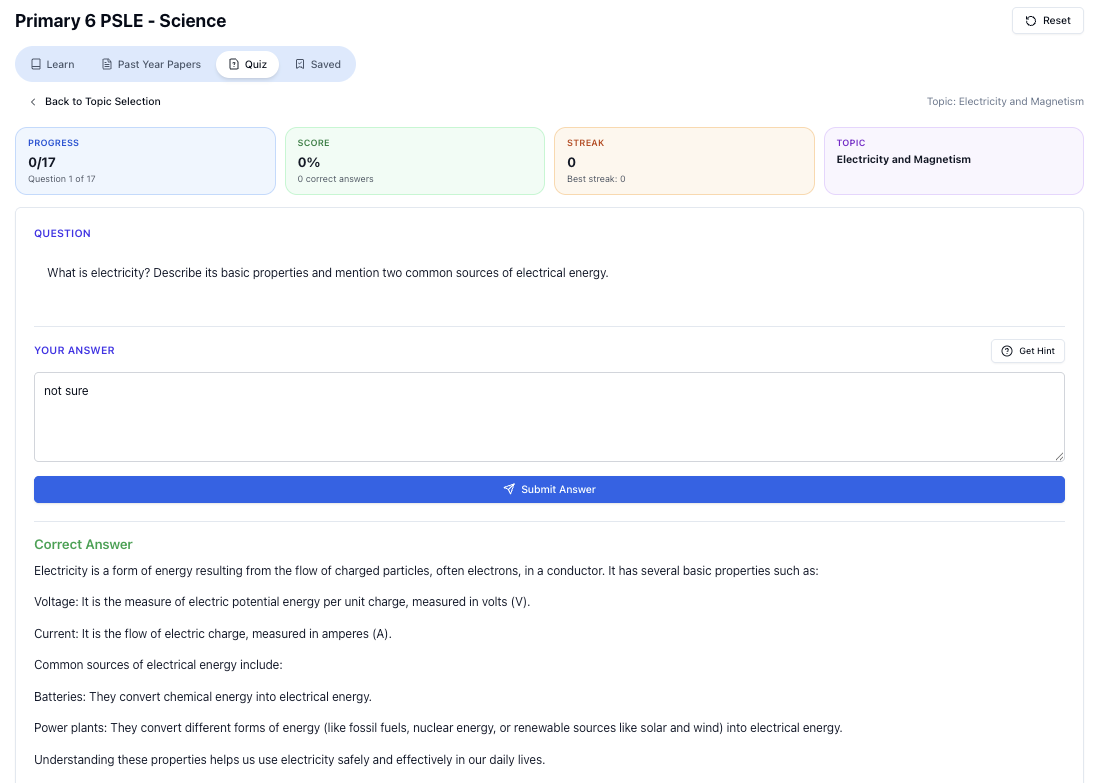
“Doing Secondary Science? Pick a topic and practise like it’s a real exam — with clear answers right after.”
👉 Try Tutorly now and start a Science topic in seconds.
![Secondary Science topics you can practise on Tutorly.sg]
A reversible reaction is studied at a fixed temperature. Initially, only A is present. The concentration–time graph shows that equilibrium is reached after 10 minutes.
(a) Explain, in terms of rates, what is happening between 0–10 minutes and after 10 minutes.
(b) Sketch and explain how the graph would change if a catalyst is added at t = 5 minutes.
(c) The value of for the reaction is 4.0. State and explain whether the value of changes when a catalyst is added.
You’ll need to combine:
- Dynamic equilibrium concepts
- Kinetics (rate of reaction)
- Effect of catalyst on rate vs equilibrium
Hard Variant 2: Organic + Spectroscopy
A compound X, CHO, is known to be an ester.
(a) Suggest two possible structural isomers of X that are esters.
(b) X is found to react with aqueous sodium hydroxide to give a single organic product Y. Deduce the structure of X and Y, and write an equation for the reaction.
(c) The infrared spectrum of X shows a strong absorption around 1740 cm and no broad peak around 3200–3600 cm. Explain how this supports your structure.
This forces you to think about:
- Functional groups
- Reaction of esters with alkali
- IR spectroscopy interpretation
Type your full answer and ask Tutorly.sg:
“Mark my answer like an A Level H 2 Chem marker and show me the full solution.”
Common Mistakes H 2 Chem Students in Singapore Keep Making
Whether in school, tuition, or online, I see the same errors over and over. Fixing these can easily push you up by a grade.
1. Memorising, not understanding, organic mechanisms
Students often:
- Memorise arrows without knowing why they point a certain way
- Confuse electrophiles and nucleophiles
- Forget to show charges or lone pairs
Fix:
- For each mechanism, ask:
- Who is electron-rich? Who is electron-poor?
- Why does the nucleophile attack that carbon?
- What stabilises the intermediate?
Use Tutorly.sg to:
“Explain the SN 1 mechanism for 2-bromopropane, step by step, and compare it with SN 2. Then give me 3 short questions to test if I really understand it.”
2. Weak explanation skills for equilibrium and energetics
Common problems:
- Writing “equilibrium shifts to the right” without stating why
- Confusing “rate” with “yield”
- Mixing up “enthalpy change” with “activation energy”
Fix:
- Practise full-sentence, 2–3 mark explanations.
- Always link:
- Condition change → effect on rate or position → final observation
You can paste your explanation into Tutorly.sg and ask:
“Rewrite my explanation to match full-mark H 2 Chem standard, and show me what I missed.”
3. Careless mole and unit errors
Things like:
- Forgetting to convert cm to dm
- Mixing up mol and mol dm
- Not checking significant figures when required
Fix:
- For every calculation question, before you start:
- Underline the units
- Convert volumes to dm immediately
After you’ve done the question, ask Tutorly.sg to:
- Check your final numerical answer
- Show the full working and highlight any unit conversions you missed
4. Ignoring weaker topics until it’s too late
Students often “postpone” topics they hate:
- Electrochemistry
- Transition metals
- Organic synthesis planning
Then they realise in JC 2 that these topics carry a lot of marks.
Fix:
-
Use tuition time and AI tutor time to specifically target your weakest topics.
-
On Tutorly.sg, you can say:
“I’m weak in electrochemistry. Give me a structured revision plan with progressive questions from easy to hard.”
And then actually work through that plan.
5. Not practising enough full-length questions
Many students do:
- One or two parts of a question
- Check answers immediately
- Never build stamina for a full 10–20 mark question
Fix:
- At least once a week , do:
- One full Paper 2-style question
- One full Paper 3-style question
Then use Tutorly.sg to:
- Compare your answers with full worked solutions
- Identify where you lose most marks (calculation steps? explanation parts? organic structure?)
How Tutorly.sg Fits into Your H 2 Chem Tuition Plan
If you’re already in JC, your schedule is probably packed: lectures, tutorials, CCAs, maybe physical tuition.
Here’s how Tutorly.sg can fit in realistically:
-
Between school and tuition
- Use it to clarify a concept you didn’t catch in lecture.
- Example: “Explain standard electrode potential for H 2 Chem in JC 2, with simple examples.”
-
Late-night homework and revision
- When you’re stuck on a tutorial question at 11pm, paste it in and get a step-by-step solution.
- Use it to check your final answers and see the full working.
-
During revision period (before promos, blocks, A Levels)
- Generate topic-specific practice questions.
- Ask for hard variants that are slightly above TYS difficulty.
- Use it to polish your explanation answers to mark-scheme standard.
Because Tutorly.sg is built for MOE syllabus and JC levels, it “thinks” in the same framework your teachers and exam setters do. And since it’s already been used by thousands of students in Singapore and even mentioned on CNA, you’re not wasting time figuring out if it’s relevant.
You can explore the AI tutor here:
👉 https://tutorly.sg/ai-tutor-singapore
Final Thoughts: Don’t Fight H 2 Chem Alone
H 2 Chemistry is tough, but it’s also very trainable.
If you:
- Learn each topic with a step-by-step approach
- Follow a clear exam strategy for Papers 1, 2, and 3
- Do worksheet practice with both standard and hard variants
- Avoid the common mistakes most JC students make
Then H 2 Chem becomes less of a mystery and more like a game you can actually win.
Focused H 2 Chem tuition helps a lot — whether that’s your school teacher, a private tutor, or an AI tutor that’s always awake when you are.
If you want a **24
Try Tutorly.sg (Singapore)
Start here: AI Tutor Singapore
Try Tutorly on the website : https://tutorly.sg/app
“Practice PSLE Science questions and get clear, step-by-step answers instantly.”
👉 Try a question now and see how fast you can improve.
Ready to practise?
If you want a Singapore-focused AI tutor you can use immediately , try Tutorly here: