Finding the best online Chemistry tutor for your O Levels in Singapore can feel stressful.
You’re juggling CCA, other subjects, maybe even tuition… and Chemistry just keeps throwing you things like moles, redox and salt preparation. On top of that, there are so many options: private tutors, Zoom classes, recorded videos, and now AI tutors like Tutorly.sg.
“Stuck on a question? See simple explanations that help you understand fast.”
👉 Give it a try and turn confusion into clarity in minutes.
This guide is written specifically for Secondary 3–4 / O Level students in Singapore. I’ll walk you through:
- How to compare different types of online Chemistry tutors
- A step-by-step tutorial for using an online tutor effectively
- An exam strategy guide tailored to O Level Chemistry
- Worksheet practice ideas (with hard variants) you can try immediately
- Common mistakes students make when choosing and using online tutors
And along the way, I’ll show you exactly how to use Tutorly.sg as your 24/7 AI Chemistry tutor, aligned to the MOE syllabus.
Why “Best Online Chemistry Tutor” Is Different For O Level Students
When you search for the best online Chemistry tutor, you probably see:
- Big tuition centres with Zoom classes
- Individual tutors doing lessons on Google Meet
- Pre-recorded video courses
- AI tutors and websites
For O Level Chemistry in Singapore, “best” should not just mean “most famous” or “most expensive”. It should mean:
-
Aligned to MOE / O Level syllabus
- Knows the exact topics: e.g. qualitative analysis, electrolysis, mole concept, rate of reaction, energy changes.
- Understands SEAB question style and common phrasing in O Level papers.
-
Able to explain why, not just what
- Not just “memorise this answer”, but “here’s the reasoning behind it”.
- Especially important for structured questions and data-based questions.
-
Gives you enough practice, with feedback
- You need exposure to basic, medium and hard variants of each topic.
- Answers must be checked quickly so you don’t repeat the same mistake for weeks.
-
Fits your schedule and attention span
- If you’re tired after school and CCA, a 2-hour Zoom class at 8pm might not be ideal.
- Sometimes 15–20 minutes of focused, targeted help is much more effective.
-
Sustainable cost
- Weekly 1-to-1 tuition can go above $300+ per month.
- For many students, a mix of school lessons, occasional human help, and a 24/7 AI tutor like Tutorly.sg is more realistic.
Comparing Your Options: Human Tutor vs AI Tutor (Like Tutorly.sg)
Here’s a simple comparison from an O Level Chem student’s point of view.
“Access more than 1000+ past year papers to practice”
👉 Start a paper today and test yourself like it’s the real exam.

1. Human Online Tutor (Zoom / Meet)
Pros:
- Can read your body language and adjust pace.
- Can focus on your school’s worksheets and teacher’s style.
- Good for students who really need someone to “nag” and keep them on track.
Cons:
- Fixed timing – if you’re tired that day, the lesson is still happening.
- Limited contact hours – you can’t ask questions at 11pm before a test.
- Quality varies a lot; not every tutor is strong in O Level exam techniques.
- More expensive, especially 1-to-1.
2. Online Group Classes
Pros:
- Cheaper than 1-to-1.
- Can be motivating if your friends join.
- Some centres are very exam-focused, with strong track records.
Cons:
- Pacing is fixed; if you’re weak in one topic, the class just moves on.
- Hard to ask many questions when there are 20–30 students.
- Usually only once a week; not ideal if you get stuck mid-week.
3. AI Tutor (e.g. Tutorly.sg)
Tutorly.sg is a 24/7 AI tutor website built specifically for Singapore students, including O Level Chemistry. It’s not a random overseas app; it’s made for MOE syllabus and has been:
- Mentioned on Channel NewsAsia (CNA)
- Used by thousands of students in Singapore
Pros:
- Always available – 11pm before your Chem test, 6am on exam day, anytime.
- You choose the topic and question; it gives step-by-step solutions to O Level style problems.
- Aligned to MOE / O Level content, so you’re not learning extra unnecessary stuff.
- Much more affordable than weekly 1-to-1 tuition.
Cons:
- It checks your final answer, then shows you how to get there – it doesn’t track every step you write.
- You need some self-discipline to keep asking good questions and practising.
Best setup for many students:
- School lessons for core content
- Occasional human help if needed
- Daily or frequent use of Tutorly.sg for practice, explanations, and last-minute questions
If you want to try it, you can go straight to:
👉 https://tutorly.sg/app
Step-by-step Tutorial: Using An Online Chemistry Tutor Effectively
Most students just “ask when stuck”. That’s okay, but not efficient. Here’s a simple system you can follow, especially if you’re using Tutorly.sg.
Step 1: Choose A Specific Topic (Don’t Be Vague)
Instead of:
“Chemistry is hard. Help.”
Be specific, like:
- “Mole concept – converting mass to moles and then to particles.”
- “Writing ionic equations for precipitation reactions.”
- “Explaining why aluminium is used for overhead electrical cables.”
On Tutorly, you select level and subject (Chemistry) first, so it already knows your syllabus. Then you can dive straight into your topic.
Step 2: Start With A Real Question
Use:
- Your school worksheet
- Ten-year-series (TYS)
- A practice paper from your teacher
- A question you typed out yourself
Example question (mole concept):
2.70 g of aluminium reacts completely with excess hydrochloric acid.
(a) Calculate the number of moles of aluminium used.
(b) Hence, calculate the number of moles of hydrogen gas formed.
(c) Calculate the volume of hydrogen gas formed at room temperature and pressure (r.t.p.).
(Given: , molar gas volume at r.t.p. )
Equation:
Step 3: Attempt First, Then Ask For Help
Don’t straightaway copy-paste the question and wait for the answer. You’ll learn much more if you:
- Try the question on your own.
- Write down your final answer clearly.
- Then ask your online tutor (or Tutorly) to check.
On Tutorly.sg:
- You enter the question and your final answer.
- Tutorly checks your answer.
- Whether you’re right or wrong, it will show you a step-by-step solution so you can see the proper method.
For the aluminium question, you’ll see steps like:
- From the equation, of gives of .
So,
- Volume at r.t.p.:
Step 4: Ask “Why”, Not Just “What”
Once you see the solution, don’t just move on. Ask follow-up questions like:
- “Why do we multiply by instead of ?”
- “How do I know when to use molar gas volume vs ?” (for O Level, you usually use molar gas volume)
- “Can you give me a similar but slightly harder question?”
Tutorly can then:
- Re-explain the concept in simpler words
- Give you another practice question of similar difficulty
- Show the step-by-step solution again
Step 5: Summarise In Your Own Words
After finishing a question, write down in your notebook:
- The key formula you used
- The pattern (e.g. mass → moles → mole ratio → volume)
- One common mistake to avoid
Example summary:
For metal + acid questions that produce gas:
- Convert mass of metal to moles.
- Use balanced equation to find moles of gas.
- Multiply by to get volume at r.t.p.
Common mistake: Forgetting to use the mole ratio from the balanced equation.
This way, your online tutor session becomes actual revision notes.
Exam Strategy Guide For O Level Chemistry (Using Online Help Smartly)
Online tutoring is most powerful when it supports a solid exam strategy. Here’s how to plan from now till your O Levels.
1. Know The Big Topics That Always Appear
For O Level Pure / Combined Chemistry, some high-yield topics include:
- Mole concept & chemical calculations
- Acid, bases & salts
- Qualitative analysis (QA)
- Electrolysis
- Redox & energy changes
- Rate of reaction
- Metals & extraction
- Organic chemistry (for Pure Chem)
Your online tutor (or Tutorly) should be very comfortable with these. When you’re choosing a human tutor, ask them:
“Can you show me how you’d teach mole concept for O Levels?”
“How do you prepare students for qualitative analysis questions?”
When using Tutorly, you can directly test it with real questions from these topics and see the step-by-step breakdown.
2. Paper 1 (MCQ) Strategy
Paper 1 is often where students lose “easy” marks due to:
- Rushing
- Misreading units
- Getting tricked by common misconceptions
Use online tutoring to:
- Practise 10–15 MCQs at a time
- After each set, ask your tutor or Tutorly to explain only the ones you got wrong
- For each wrong question, ask:
- “What concept is this really testing?”
- “What trap did I fall for?”
Over time, you’ll build a list of your personal traps, e.g.:
- Forgetting that ionic compounds conduct electricity only when molten or in solution.
- Confusing exothermic vs endothermic energy profile diagrams.
- Mixing up empirical vs molecular formula.
3. Paper 2 (Structured & Free Response) Strategy
Paper 2 is where good explanation skills and clear working matter.
Use an online tutor to:
- Practise writing full answers, not just final values.
- Get model answers and compare phrasing.
- Learn how to structure explanations.
Example: “Explain why increasing temperature increases the rate of reaction.”
A strong answer (O Level style) should mention:
- Particles gain kinetic energy
- Higher frequency of effective collisions
- More particles have energy equal to or greater than activation energy
You can ask Tutorly:
“I wrote this answer. How can I improve it to match O Level marking scheme?”
Then refine your phrasing across multiple questions.
4. Prelim & O Level Period: How To Use Online Help Daily
Near exams, you don’t need 3-hour marathons every day. You need short, focused sessions.
A simple daily plan:
- 15–20 minutes: Do 2–3 structured questions from your weakest topic
- 10 minutes: Check with Tutorly or your online tutor, review step-by-step solutions
- 5 minutes: Summarise what you did wrong and how to fix it
Repeat this for different topics each day. This is where a 24/7 AI tutor like Tutorly.sg is very useful – you can slot it in anytime you’re free, without scheduling.
Worksheet Practice (With Hard Variants You Can Try)
Here are sample practice questions you can try now. You can attempt them, then use Tutorly.sg to:
- Check your final answers
- Get step-by-step solutions
- Ask for similar questions
Topic 1: Mole Concept & Stoichiometry
Q 1 (Basic)
Magnesium reacts with dilute hydrochloric acid according to the equation:
(a) Calculate the number of moles of magnesium in 6.0 g of magnesium.
(b) Calculate the number of moles of hydrogen gas formed.
(Relative atomic mass: )
Q 2 (Hard Variant)
Calcium carbonate is heated strongly to form calcium oxide and carbon dioxide:
A student heats of impure calcium carbonate and obtains of carbon dioxide.
(Relative formula mass: , )
(a) Calculate the number of moles of produced.
(b) Calculate the number of moles of that actually decomposed.
(c) Calculate the mass of pure that decomposed.
(d) Hence, calculate the percentage purity of the original sample of calcium carbonate.
This kind of purity question is very common in O Levels and often appears in different disguises. After solving, ask Tutorly:
“Can you show me another purity question, but with a different reaction?”
Topic 2: Qualitative Analysis
Q 3 (Medium)
A colourless solution X gives a white precipitate with aqueous sodium hydroxide that is insoluble in excess. The same solution gives a white precipitate with aqueous ammonia that is insoluble in excess.
(a) Suggest the cation present in solution X.
(b) State one possible anion present in solution X and describe a test for it.
Q 4 (Hard Variant – Data Combination)
A solid Y is soluble in water to form a colourless solution. The following tests were carried out:
- To a portion of the solution, aqueous sodium hydroxide is added dropwise until in excess. A light blue precipitate is formed, which dissolves in excess to give a deep blue solution.
- To another portion of the solution, dilute nitric acid is added, followed by aqueous silver nitrate. A white precipitate is formed.
(a) Identify the cation in Y.
(b) Identify the anion in Y.
(c) Suggest the chemical name and formula of Y.
This question combines cation and anion identification – typical of O Level structured questions. After answering, you can ask Tutorly:
“Explain clearly why the cation is [your answer], step by step.”
Topic 3: Electrolysis
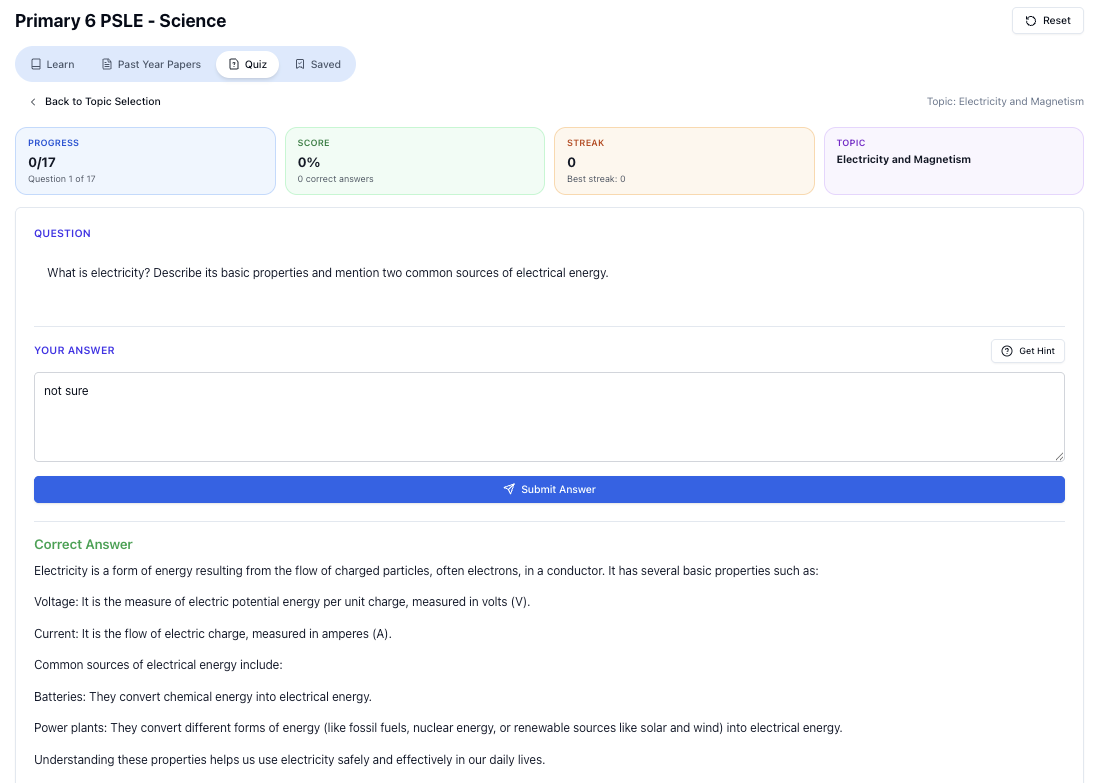
“Doing Secondary Science? Pick a topic and practise like it’s a real exam — with clear answers right after.”
👉 Try Tutorly now and start a Science topic in seconds.
![Secondary Science topics you can practise on Tutorly.sg]
Q 5 (Basic)
Dilute sodium chloride solution is electrolysed using graphite electrodes.
(a) Name the ions present in the solution.
(b) State the product formed at the cathode.
(c) State the product formed at the anode.
Q 6 (Hard Variant – Reasoning)
Concentrated aqueous sodium chloride is electrolysed using graphite electrodes. The products formed are hydrogen gas, chlorine gas and sodium hydroxide solution.
(a) Explain why hydrogen gas, and not sodium metal, is produced at the cathode.
(b) Explain why chlorine gas is produced at the anode instead of oxygen gas.
(c) State one use for each of the products formed.
Here, you’re tested on electrochemical series and preferential discharge. After attempting, ask Tutorly:
“Can you give me a summary of how to decide which ion is discharged at each electrode for O Level?”
Topic 4: Rate Of Reaction
Q 7 (Medium)
Marble chips (calcium carbonate) react with dilute hydrochloric acid to produce carbon dioxide gas. Describe and explain the effect of:
(a) increasing the temperature of the acid
(b) decreasing the size of the marble chips
on the rate of reaction.
Q 8 (Hard Variant – Graph & Explanation)
A student investigates the reaction between excess marble chips and dilute hydrochloric acid at two different temperatures, and . The volume of carbon dioxide produced is measured over time.
(a) Sketch two curves on the same set of axes to show how the volume of carbon dioxide changes with time at the two temperatures. Label your curves.
(b) Explain why the curve at is steeper at the start.
(c) Explain why both curves eventually level off at the same volume.
This question tests your graph interpretation and kinetic theory explanation. After trying, you can ask Tutorly to:
- Check whether your explanation covers all key points (kinetic energy, frequency of effective collisions, same amount of reactants).
Topic 5: Organic Chemistry (For Pure Chemistry)
Q 9 (Medium)
Ethene reacts with steam in the presence of a catalyst to form ethanol.
(a) Name this type of reaction.
(b) Write a chemical equation for this reaction.
(c) State one condition needed for this reaction.
Q 10 (Hard Variant – Isomers & Properties)
Two organic compounds, A and B, have the same molecular formula .
(a) Draw the displayed formulae of A and B.
(b) State the type of isomerism shown by A and B.
(c) Describe one chemical test that can be used to distinguish between A and B, and state the expected observations.
These are typical higher-order organic questions where many students lose marks. After solving, you can ask Tutorly:
“Show me a step-by-step explanation of how to distinguish between ethanol and dimethyl ether using O Level methods.”
Common Mistakes When Choosing & Using Online Chemistry Tutors
Mistake 1: Choosing A Tutor Not Familiar With MOE / O Level Style
Some online tutors (especially overseas ones) may:
- Teach content not in the O Level syllabus
- Use A Level or IGCSE style questions that are not aligned
- Miss out on local exam trends (e.g. common practical or QA questions)
Always check:
- Do they mention O Levels / MOE syllabus clearly?
- Can they show you solutions to actual O Level / prelim questions?
With Tutorly.sg, this is already taken care of — it’s built specifically for Singapore MOE students, from Primary to JC, and includes O Level Chemistry content.
Mistake 2: Treating Online Tutoring As A “Magic Fix”
Even the best tutor (human or AI) can’t help if:
- You don’t attempt questions yourself
- You only start using it one week before exams
- You keep asking for answers without understanding the method
To avoid this:
- Always try first, then ask for help.
- Use the step-by-step solutions to learn the method, not just copy.
- Re-do similar questions until you can solve them without help.
Mistake 3: Not Reviewing Your Own Errors
Many students:
- Look at the solution
- Say “ok I understand”
- Then repeat the same mistake in the next test
Instead, create a “Mistake Log”:
For each question you get wrong:
- Write the topic (e.g. Mole concept – limiting reagent)
- Copy the question (shortened)
- Note your wrong method
- Write the correct method and why you were wrong
You can use Tutorly to help fill in the correct method and explanation. Review this log weekly – these are the questions most likely to appear again in slightly different forms.
Mistake 4: Ignoring Time Management
Online help can be so convenient that you end up:
- Spending 1 hour on a single question
- Going too deep into one topic and ignoring others
For O Levels, you need breadth and depth.
To manage time:
- Decide in advance: “I’ll spend 30 minutes on Mole concept today.”
- Set a timer. When time’s up, move to another topic.
- If a question is too hard, ask Tutorly for a simpler version first, then build up.
Mistake 5: Not Using The Right Tool For The Right Task
Use different tools for different needs:
- School teacher / notes – main content and official expectations
- Human tutor – motivation, personalised feedback, long-term guidance
- Tutorly.sg – fast, targeted help anytime, especially for:
- Step-by-step solutions
- Checking your answers
- Getting extra practice questions
- Clarifying confusing concepts right away
When you combine them properly, your Chemistry preparation becomes much more efficient.
Final Thoughts: How To Actually Get Better At O Level Chemistry
To truly benefit from the best online Chemistry tutor for O Levels, focus on this simple cycle:
- Learn a concept – from school / notes / teacher
- Practise questions – from worksheets, TYS, online
- Check & understand solutions – with Tutorly or a human tutor
- Record your mistakes – so you don’t repeat them
- Repeat with harder variants – until you can solve them confidently
If you want an online tutor that:
- Is aligned to the Singapore MOE O Level syllabus
- Is available 24/7 (even late at night before your test)
- Has already been used by thousands of students in Singapore
- Has been mentioned on CNA
Then it’s worth trying Tutorly.sg.
You can start using the AI tutor directly here:
👉 https://tutorly.sg/app
Use it to:
- Check your Chemistry answers
- Get step-by-step solutions
- Practise harder worksheet variants
- Clarify doubts immediately, instead of waiting for the next lesson
With consistent practice and the right support, O Level Chemistry doesn’t have to stay your “killer subject”. You can turn it into a paper where you walk into the exam hall knowing exactly what to expect — and how to handle it.
“Practice PSLE Science questions and get clear, step-by-step answers instantly.”
👉 Try a question now and see how fast you can improve.
Ready to practise?
If you want a Singapore-focused AI tutor you can use immediately , try Tutorly here: