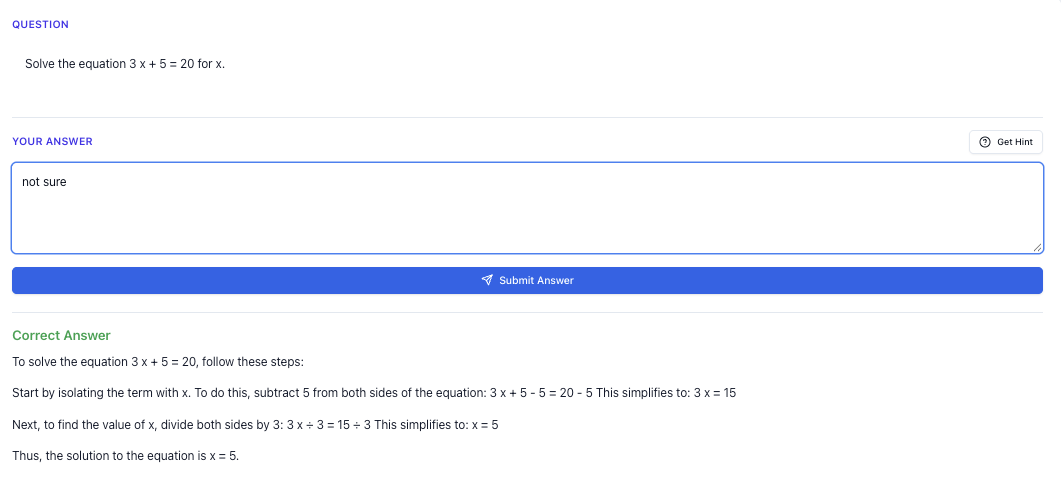
If you’re in JC, you already know: A Level Chemistry is no joke.
Between lecture tests, SPA-style questions , and promo/prelim pressure, it’s normal to start Googling “best JC chemistry tuition” at 1am and wondering if you’re the only one falling behind.
“Stuck on a question? See simple explanations that help you understand fast.”
👉 Give it a try and turn confusion into clarity in minutes.
You’re not.
This guide is written for you if:
- You’re deciding whether to join a big-name JC Chem tuition centre
- You’re already in tuition but still stuck on application questions
- You’re considering cheaper or more flexible options like AI help
I’ll walk you through:
- How to compare the main types of JC Chemistry tuition in Singapore
- Where Tutorly.sg fits in (and when it can actually be better than tuition)
- A step-by-step tutorial on tackling typical A Level Chem question types
- A realistic exam strategy guide
- Worksheet-style practice, including hard variants
- The common mistakes JC students make with Chem – and how to fix them
Tutorly.sg has already been used by thousands of students in Singapore and has been featured on Channel NewsAsia (CNA), so I’ll be very direct about when it helps more than a human class, and when you might still want a physical tutor.
Comparing the Best JC Chemistry Tuition Options in Singapore
When people say “best JC Chemistry tuition”, they usually mean one of these:
- Big branded group tuition centres
- Neighbourhood/small group tutors
- Private 1-to-1 home tutors
- Online/AI-based help like Tutorly.sg
Let’s compare them honestly, from a JC student’s point of view.
1. Big branded JC Chemistry tuition centres
Think of the well-known names with:
- Fixed weekly classes
- Printed notes and topical summaries
- Past-year prelim question booklets
- Sometimes recorded lessons
Pros:
- Structured coverage of the full H 1/H 2 syllabus
- Strong focus on exam trends and “what Cambridge likes”
- You get exposed to challenging prelim questions from top JCs
- Friends may be there – easier to stay motivated
Cons:
- Fixed timing – if you have CCA / council / competitions, you’ll miss lessons
- Class size can be big , so you may be shy to ask “basic” questions
- If you’re weak in foundations (e.g. mole concept, redox), the pace may be too fast
- Travelling time adds up, especially if you stay far from the centre
Best for you if:
- You want a weekly rhythm and someone to “drag” you through the syllabus
- You’re already around average and want to push to B/A
- You like having printed notes and a fixed routine
2. Neighbourhood / small group JC Chem tutors
These are usually ex-teachers or experienced tutors teaching in smaller groups .
Pros:
- Smaller groups = easier to ask questions
- Tutor can adjust pace to your school’s scheme of work
- Often cheaper than big branded centres
- More flexible about rescheduling
Cons:
- Quality varies a lot – not all are familiar with the latest A Level question styles
- Materials may not be as polished or extensive
- If group is mixed , teaching might feel scattered
Best for you if:
- You want more attention than large centres, but still like group learning
- You’re okay to try 1–2 lessons first and see if the fit is good
3. 1-to-1 JC Chemistry home tuition
The classic option.
Pros:
- Fully personalised – can re-teach Sec 3/4 concepts if needed
- You can focus on your school’s tutorials and tests
- Good for students who are very behind or very shy
- Flexible timing (within reason)
Cons:
- Most expensive option
- Quality depends entirely on that one tutor
- If you’re tired or unprepared, a 2-hour session can feel wasted
- Some students become over-dependent and stop trying questions themselves
Best for you if:
- You’re failing badly and need someone to rebuild foundations
- Or you’re aiming for A in H 2 Chem for Medicine/Pharmacy and want to polish every topic
4. Online / AI-based help – where Tutorly.sg fits in
This is where many JC students are still a bit blur: not all “AI help” is the same.
Tutorly.sg is a 24/7 AI tutor website built specifically for Singapore students, aligned to the MOE syllabus from Primary 1 to JC 2. It’s not a random overseas chatbot, and it’s not a mobile app – you just use it through the website.
Relevant links for you:
- Main AI tutor page (how it works): https://tutorly.sg/ai-tutor-singapore
- Go straight to using it: https://tutorly.sg/app
For JC Chemistry, you can:
- Type or paste your exact exam question
- Choose your level (e.g. JC 2 H 2 Chemistry)
- Get a final answer + step-by-step working aligned to A Level standards
- Ask follow-up questions like “why is this nucleophile stronger?” or “compare with SN 2”
What AI (like Tutorly.sg) is good at:
- Being awake at 1–3am when you’re rushing tutorials
- Repeating explanations as many times as you want, without judgment
- Generating similar practice questions when you say “give me a harder one”
- Helping you check if your final answer is correct, then showing the steps
What it’s NOT doing:
- It doesn’t “see” your scribbled working and mark every step
- It won’t replace school practicals or real lab work
- It won’t magically study for you – you still need to try questions first
Best for you if:
- You already have school lessons, and maybe tuition, but still get stuck on individual questions
- You want something cheaper and more flexible than extra tuition classes
- You like learning at your own pace and asking “why” without feeling paiseh
Realistic combo that works for many JC students:
- Option A: Big/Small group Chem tuition for structure + Tutorly.sg for daily homework help and last-minute revision
- Option B: No tuition, just school + consistent use of Tutorly.sg for practice, explanations, and clarifying doubts
Step-by-step Tutorial: How to Tackle Common A Level Chem Question Types
Instead of just talking about tuition, let me walk you through how a good tutor (or Tutorly.sg) would guide you through typical A Level question types.
“Access more than 1000+ past year papers to practice”
👉 Start a paper today and test yourself like it’s the real exam.

1. Structured calculation questions (Mole concept, Energetics, Equilibria)
Example style :
A sample of 2.50 g of a monoprotic organic acid HA is dissolved in water and made up to 250 cm³. 25.0 cm³ of this solution requires 22.4 cm³ of 0.100 mol dm⁻³ NaOH for complete neutralisation.
(a) Calculate the concentration of HA in the original solution.
(b) Calculate the relative molecular mass, , of HA.
Step-by-step approach:
-
Identify what’s happening
- Monoprotic acid + NaOH → neutralisation
- 1 mol HA reacts with 1 mol NaOH
-
Calculate moles of NaOH used in titration
- Volume in dm³:
- Moles:
-
Use mole ratio to find moles of HA in 25.0 cm³
- 1:1 ratio ⇒
-
Scale up to 250 cm³
- 25.0 cm³ → 2.24 × 10⁻³ mol
- 250 cm³ is 10 times, so moles in full solution:
-
Concentration of HA (part a)
- Volume = 0.250 dm³
-
Find using mass and moles (part b)
- Mass given = 2.50 g
- Moles in that mass = 2.24 × 10⁻² mol
How Tutorly.sg helps here:
You can:
- Paste the question into https://tutorly.sg/app
- Get the final answers 0.0896 mol dm⁻³ and
- Then see the full working step-by-step, similar to above
- Ask: “Why multiply by 10 in step 4?” or “Explain again the 1:1 ratio”
2. Organic mechanism questions (Nucleophilic substitution, Electrophilic addition)
Example style:
Chloroethane reacts with aqueous sodium hydroxide to form ethanol.
(a) Name the type of reaction.
(b) Outline the mechanism of this reaction.
Step-by-step thinking:
-
Identify functional groups:
- Chloroethane: halogenoalkane
- Reagent: aqueous NaOH (OH⁻ as nucleophile)
-
Recognise typical mechanism:
- Primary halogenoalkane + OH⁻ → nucleophilic substitution
-
Mechanism outline:
- Lone pair on OH⁻ attacks the C atom bonded to Cl from the opposite side
- Simultaneous breaking of C–Cl bond
- Transition state with partial bonds
- Cl⁻ leaves as leaving group
-
Marking scheme usually wants:
- Curly arrow from lone pair on O of OH⁻ to C
- Curly arrow from C–Cl bond to Cl
- Product: CH₃CH₂OH + Cl⁻
If you ask Tutorly.sg, you’ll get:
- The name of the reaction: nucleophilic substitution ()
- A step-by-step verbal description of the mechanism (since it only outputs text)
- Clarifications like “compare vs ” or “what about tertiary halogenoalkanes?”
3. Data-based / planning questions (Energy changes, Kinetics, Equilibrium)
These often show tables or graphs and ask you to:
- Interpret trends
- Suggest explanations based on collision theory / Le Chatelier’s principle
- Propose experiments or improvements
Example style:
The rate of decomposition of hydrogen peroxide was measured at different temperatures. The following data were obtained…
(table given)
(a) Plot a suitable graph to determine the activation energy, .
(b) Explain how the graph can be used to determine .
Here, you need to know:
- Use Arrhenius equation:
- Take vs
- Gradient =
Tutorly.sg can:
- Walk you through which graph to plot and why
- Show the algebra steps from Arrhenius equation to the linear form
- Remind you of units and common traps (e.g. using °C instead of K)
Exam Strategy Guide for JC Chemistry (H 1 & H 2)
Tuition (even the best one) won’t help if your exam strategy is off. Here’s how to think about A Level Chem from now till your papers.
1. Know the paper structure (H 2 focus)
Typical H 2 Chemistry (Paper codes may differ slightly by year, but structure is similar):
- Paper 1 (MCQ) – 40 marks
- Paper 2 (Structured) – ~72 marks
- Paper 3 (Free response) – ~80 marks
H 1 has fewer papers, but the style is similar: MCQs + structured questions.
You need different strategies for each.
Paper 1 (MCQ)
- Aim to finish in 35–40 mins, then use remaining time to check
- Don’t spend 5 minutes stuck on one question; circle and move on
- Build a personal MCQ notebook of “tricky concepts” (e.g. wording on buffers, Kc vs Q)
Use Tutorly.sg to:
- Generate similar MCQs to any topic you’re weak in
- Ask for explanations for each option, not just the correct one
Paper 2 & 3 (Structured + Free response)
- Time yourself: roughly 1.5 min per mark as a guideline
- Underline command words: “Explain”, “State”, “Deduce”, “Suggest”
- For long questions, quickly jot a mini-plan
Use Tutorly.sg when practicing:
- Type: “Mark this like an A Level question; show me the full working and what I missed.”
- It can’t literally mark each line of your script, but it can:
- Show a model answer
- Highlight key points that should appear
- Help you compare your answer with the ideal structure
2. Topic-by-topic strategy (what JC Chem tuition often does)
Most top tuition centres follow a similar progression:
- Physical Chemistry foundations
- Atomic structure, bonding, energetics, kinetics, equilibrium
- Inorganic Chemistry
- Periodicity, Group 2, Group 17, transition elements
- Organic Chemistry
- Functional groups, mechanisms, spectroscopy, synthesis
You can copy this structure even without tuition:
- Use your lecture notes + Ten Year Series (TYS) + Tutorly.sg
- For each topic:
- Revise key concepts and definitions
- Do 5–10 TYS questions of increasing difficulty
- When stuck, ask Tutorly.sg for step-by-step help
- Write down 1–2 “golden mistakes” you made so you don’t repeat them
3. How to use Tutorly.sg effectively with (or without) tuition
If you’re already in tuition:
- Before class:
- Skim the topic
- Ask Tutorly.sg 1–2 conceptual questions so you’re not lost in class
- After class:
- Redo similar questions without looking at the solution
- Check with Tutorly.sg only after you’ve tried
If you’re not in tuition:
- Treat Tutorly.sg as your on-demand tutor:
- After school tutorials, paste any question you couldn’t do
- Ask it to explain wrong options in MCQs, not just the correct one
- Ask for “a slightly harder version of this question” to stretch yourself
Main point: Don’t just use it to copy answers. Use it to understand patterns.
Worksheet Practice (With Hard Variants)
Let’s go through some practice-style questions you can try right now. I’ll give you the question, then a brief outline of the solution. For full step-by-step workings, you can paste them into https://tutorly.sg/app and ask for detailed solutions.
Section A: Core Practice Questions
“Doing Secondary Science? Pick a topic and practise like it’s a real exam — with clear answers right after.”
👉 Try Tutorly now and start a Science topic in seconds.
![Secondary Science topics you can practise on Tutorly.sg]
Q 1: Equilibrium (H 2 level)
Nitrogen dioxide, NO₂, dimerises to form dinitrogen tetroxide, N₂O₄, according to the equation:
At 298 K, 1.00 mol of NO₂ is placed in a 2.00 dm³ vessel. At equilibrium, 0.60 mol of N₂O₄ is present.
- Calculate the equilibrium concentrations of NO₂ and N₂O₄.
- Calculate the equilibrium constant, , at this temperature.
Outline of solution:
- Let initial moles of NO₂ = 1.00 mol; N₂O₄ = 0
- At equilibrium, moles of N₂O₄ = 0.60 mol
- This comes from 2 × 0.60 = 1.20 mol of NO₂, but we only had 1.00 mol… so check logic.
Careful: Because 2 NO₂ → N₂O₄, forming 0.60 mol N₂O₄ consumes 1.20 mol NO₂. That’s impossible from 1.00 mol. So something’s off.
This is exactly the kind of trap A Level questions love.
Better assumption: Let x = moles of N₂O₄ formed. Then:
- NO₂ consumed = 2 x
- At equilibrium, moles of N₂O₄ = x
- Given x = 0.60 mol
- NO₂ at equilibrium = 1.00 − 2 = 1.00 − 1.20 = −0.20 mol (impossible)
So the data implies that some NO₂ was already present as N₂O₄ initially or the question is incomplete. If this was a real exam question, more data would be given, e.g. total pressure or initial mixture.
If you paste a corrected version (or a full question) into Tutorly.sg, it will:
- Help you set up the ICE table properly
- Guide you through concentration calculations
- Compute correctly
For now, focus on the method:
- Use ICE table (Initial, Change, Equilibrium)
- Convert moles to concentration by dividing by volume
- Substitute into:
Q 2: Organic – Identifying functional groups (H 2)
A compound X has the molecular formula C₄H₈O₂. It is known that:
- X reacts with aqueous sodium carbonate to produce effervescence.
- X does not react with 2,4-DNPH reagent.
- On heating with acidified potassium dichromate(VI), X does not change the colour of the reagent.
- Deduce the functional group(s) present in X.
- Suggest a possible structural formula for X.
Outline of solution:
- Effervescence with Na₂CO₃ → presence of carboxylic acid (CO₂ gas produced)
- No reaction with 2,4-DNPH → no aldehyde or ketone
- No oxidation with acidified K₂Cr₂O₇ → no primary or secondary alcohol, no aldehyde
So X is likely a carboxylic acid without any oxidisable –OH or carbonyl beyond the –COOH.
C₄H₈O₂ with a carboxylic acid: general formula for carboxylic acids. For n=4, C₄H₈O₂ fits.
Possible structure: butanoic acid, CH₃CH₂CH₂COOH.
Tutorly.sg can:
- Check your proposed structure
- Suggest isomers and test each against the given reactions
- Explain why other isomers (e.g. methyl propanoate) don’t fit the tests
Section B: Hard Variants (Stretch Yourself)
These are the kind of questions top JCs like to put in prelims.
Q 3 (Hard): Buffer + pH calculation (H 2)
A buffer solution is prepared by dissolving 0.200 mol of ethanoic acid, CH₃COOH, and 0.150 mol of sodium ethanoate, CH₃COONa, in water to make 1.00 dm³ of solution. The of ethanoic acid is 4.76.
- Calculate the pH of the buffer solution.
- 0.0100 mol of HCl is added to 0.100 dm³ of this buffer. Assuming volume change is negligible, calculate the new pH.
Outline of solution:
-
Initial buffer pH (using Henderson–Hasselbalch):
So pH ≈ 4.76 − 0.13 ≈ 4.63.
-
After adding 0.0100 mol HCl to 0.100 dm³ of buffer:
- Moles of acid in 0.100 dm³: mol
- Moles of salt in 0.100 dm³: mol
- HCl reacts with salt (the base component):
- New moles:
- Salt: 0.0150 − 0.0100 = 0.0050 mol
- Acid: 0.0200 + 0.0100 = 0.0300 mol
- Concentrations :
- $[\text{acid}] = \dfrac{0.
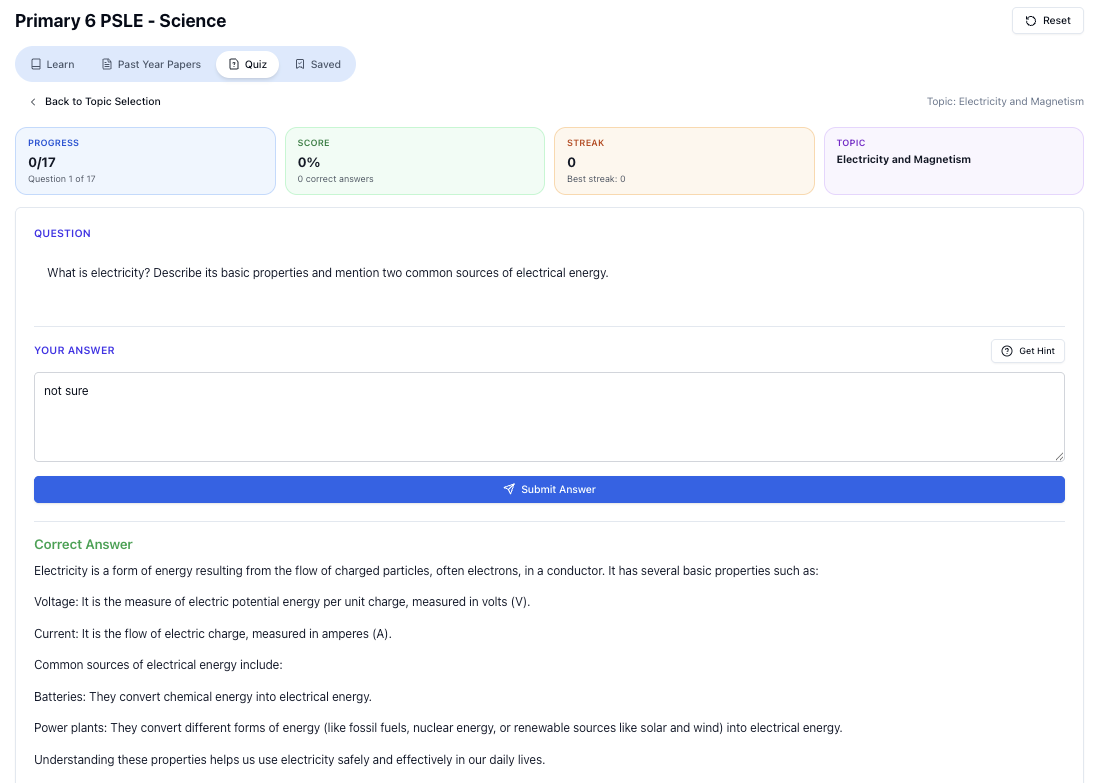
“Practice PSLE Science questions and get clear, step-by-step answers instantly.”
👉 Try a question now and see how fast you can improve.
Ready to practise?
If you want a Singapore-focused AI tutor you can use immediately , try Tutorly here: